Researchers demonstrate versatility of solid-state protein sensor

A novel type of sensor, based on nanometer-scale pores in a semiconductor membrane, is a step closer to practical use in applications such as analyzing the protein contents of a single cell. Researchers pioneering single-molecule nanopore sensor technology at the Technische Universität München (TUM) have shown its potential through a succession of experiments over the past few years. Now, in collaboration with biochemists at Goethe University Frankfurt, they have been able to advance this effort past what had been a sticking point: enhancing the selectivity of the sensor while maintaining its sensitivity to single molecules. They report the latest results in Nature Nanotechnology.
Research aimed at fabricating and controlling diagnostic devices on the scale of biological molecules has advanced to the point where several different approaches are seen as competitive for practical application in the relatively near future. The quest is not to create a universal, broad-spectrum sensor, but rather a technology for detecting and identifying single molecules of specifically targeted proteins – without the need to modify them by adding biochemical "labels." Such technology could be used, for example, to accelerate the translation of insights from genomics into working knowledge of proteomics. This is of vital interest for medical and pharmaceutical research as well as for more fundamental biological studies. Another often-mentioned potential application is detection of biological warfare agents.
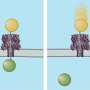
One of the most promising approaches is based on engineering a molecular "checkpoint," a monitored passageway between two salt solutions separated by an artificial membrane. This so-called nanopore is sized and shaped so that molecules must pass through one at a time. A molecule's presence in the pathway produces measurable changes in electrical conductivity across the nanopore, and further refinements can fine-tune such a detector to make it a highly specific "identity check" for targeted molecules. What the Munich-Frankfurt collaboration has enabled is a robust and reversible method for doing this fine tuning: equipping an inorganic nanopore sensor with biological functionality by anchoring molecular receptors inside it. The teams were led by Dr. Ulrich Rant, of TUM's Walter Schottky Institute and the TUM Institute for Advanced Study, and Prof. Dr. Robert Tampé of the Institute of Biochemistry at Goethe University Frankfurt and the Cluster of Excellence Frankfurt (CEF) Macromolecular Complexes.
"What I find amazing about nanopores," Rant says, "is their cost-effectiveness: You can already run single-molecule experiments with equipment worth only a couple thousand euros. If the technology is commercialized further, I am sure that instruments could be sold for the price of a personal computer, potentially allowing people to detect single molecules at home."
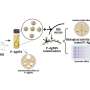
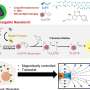
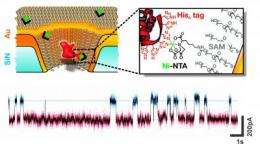
Fabrication of these experimental devices began with a substrate of silicon nitride just 50 nanometers thick; thus the membrane was literally a semiconductor chip (though not, of course, an integrated circuit). Standard tools of the microelectronics industry, electron-beam lithography and reactive ion etching, were used to pierce the silicon nitride membrane with nanopores 20 to 50 nm in diameter. Vapor deposition was used to coat the pores with titanium and gold, and the metal in turn received a self-assembled chemical coating one molecular layer thick. The resulting nanopores had a tapered conical shape with a diameter at the narrow end of around 25 nm; anchored in the surface layer were a small number of biochemical receptors designed to capture and immobilize the protein that would give the sensor its biological functionality. For these experiments, the chips were mounted in a measurement chamber with an electrolyte solution on either side; protein molecules added to one side of the chamber were urged through the nanopore by an electric charge. Traces of changes in electric current yielded real-time measurements of single molecules as they passed through the nanopores – or lingered while they interacted with the immobilized protein – and these data in turn provided positive identification of target molecules.

The researchers were able to detect recombinant histidine-tagged proteins, and also to discriminate between subclasses of natural IgG antibodies (from rats and hamsters). In the past, this level of selectivity could only be achieved at the expense of single-molecule sensitivity. The results reported in Nature Nanotechnology show that this obstacle can be overcome, and more – that the approach is extremely versatile. As Rant explains, "The method we employed to anchor single proteins inside the pore is quite generic and works for many available recombinant proteins. Hence it is easy to endow the solid-state nanopore with the desired biological functionality. Simply select a suitable protein, which specifically recognizes target proteins of interest, and use it to probe proteins as they pass by."
"The future remains exciting," Tampé says, "because nature is way ahead of us in terms of selectivity and specificity. Therefore further improvements are needed in the field of artificial sensory systems at the molecular level. The collaboration between the Munich and Frankfurt teams represents an important step in biosensory and nanodiagnostic technology on the level of single molecules." Rant adds, "Most of the recent nanopore research has been directed toward DNA detection and sequencing. I hope our results show that nanopores have the potential to become important tools for protein research too. And who knows? Maybe we'll soon see the introduction of protein nanopore sensors as diagnostic instruments to detect low-abundance disease markers in patients' samples."
More information: Stochastic sensing of proteins with receptor-modified solid-state nanopores Ruoshan Wei, Volker Gatterdam, Ralph Wieneke, Robert Tampé, and Ulrich Rant, Nature Nanotechnology, March 11, 2012. DOI: 10.1038/NNANO.2012.24
Journal information: Nature Nanotechnology
Provided by Technische Universitaet Muenchen