Results of landmark study of HIV vaccine published in the Lancet
Results from the Step study, a test-of-concept efficacy study of a Merck & Co., Inc. HIV vaccine candidate, were published online today in two papers in The Lancet. These analyses of the Step study are being conducted, presented and published to inform the continued search for an effective HIV vaccine.
The two analyses, one of the clinical results of the study and the other of study participants' immune responses to the vaccine, did not produce a clear answer to the question of why this particular vaccine candidate did not lower the risk of acquiring HIV or reduce the severity of HIV infection. The primary results of the Step study were first released in September 2007 and presented at several scientific conferences in 2007 and 2008.
The study was co-sponsored by Merck & Co., Inc., which developed the vaccine, and the National Institute of Allergy and Infectious Diseases (NIAID), part of the U.S. National Institutes of Health through the HIV Vaccine Trials Network (HVTN).
The results of Step showed that the vaccine did not prevent infection in those not previously infected with HIV, nor did the vaccine reduce the amount of virus in those study participants who became infected with HIV through exposure from an infected person while in the trial. Researchers also found that the likelihood of becoming HIV positive was highest among men who received the vaccine who were uncircumcised and had pre-existing immunity to adenovirus type 5 (Ad5), the specific cold virus used as a carrier for the vaccine, compared to those that received placebo, though the increased likelihood of becoming HIV positive may not have been limited to those subgroups.
"This trial stands as a landmark clinical HIV vaccine investigation that has profoundly informed the entire HIV vaccine research field," said Larry Corey, M.D., principal investigator of the HVTN. "Lessons learned from this trial will guide future vaccine development, and we are indebted to the many investigators and thousands of study participants for their contribution to the search for an effective HIV vaccine."
"As disappointing as the outcome of the Step study was, we will continue to follow the study participants and conduct additional analyses as part of Merck's commitment to a comprehensive approach to address the complex challenges of the AIDS pandemic," said Peter S. Kim, Ph.D., President, Merck Research Laboratories. "Merck has invested decades of time, and the expertise of hundreds of scientists into creating solutions to combat HIV. We are committed to the fight against HIV and AIDS for the long haul on many fronts."
"The development of a safe and effective HIV vaccine requires collaboration that integrates innovative ideas and cutting-edge technologies," said Alan Bernstein, Ph.D., Executive Director, Global HIV Vaccine Enterprise. "The Step study partnership among Merck, HVTN, and NIAID exemplifies the synergistic effort that the world needs if we are to stop this virus."
Immune Response to Vaccine Did Not Result in Protection from Infection or Lowering of Viral Load among Those Who Became Infected
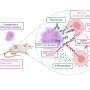
In the comprehensive analyses of immune responses to the vaccine, published for the first time today, the researchers analyzed study participants' blood samples to try to assess why the vaccine did not prevent infection and why some study participants who received the vaccine were more likely than others to develop HIV infection.
The vaccine was effective at producing an immune response: 77 percent of those vaccinated who later developed HIV infection while in the study had generated HIV-specific T-cells prior to infection. In addition, among those who received the vaccine, no major differences were found in the HIV-specific immune responses in those who developed HIV infection during the trial compared to those who did not.
Among those participants who became infected, vaccination was not associated with an effect on viral load; similar levels of circulating virus were detected among vaccine and placebo recipients.
The authors believe these findings mean that this type and level of production of an HIV-specific T-cell immune response alone may not be sufficient to prevent infection. Study participants will be followed through 2009 and further analyses are being conducted, including extensive laboratory studies to determine whether the genetic variation of HIV contributed to the vaccine's lack of effectiveness, and immunologic studies to define whether there are specific immune responses that could predict vaccine efficacy.
"If T-cell immunity is critical to produce protection against HIV, the findings from Step suggest that future candidate vaccines must elicit responses that are more broadly reactive or qualitatively different from the immune responses elicited by the Merck vaccine candidate in this trial," said Susan Buchbinder, M.D., San Francisco Department of Public Health. "It remains possible, too, that immune responses produced by T-cell based vaccines alone may not be sufficient to protect against HIV infection or disease. We will continue to explore all results of the Step study to help inform the continued search for a vaccine."
Vaccine Was Developed over Ten Years at Merck and Studied Around the World
The vaccine was created by Merck Research Laboratories and had been in development at Merck for more than a decade. The Merck adenovirus-based vaccine used a cell-mediated immune response approach; it was hypothesized that the HIV genes in the vaccine would stimulate the body to generate an HIV-specific immune response through the body's own CD8 T-cells, which become programmed to recognize and kill HIV-infected cells. Because the vaccine did not contain live HIV and contained only three HIV genes, volunteers could not become infected with HIV from vaccination.
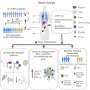
The Step study (HVTN 502, Merck V520 Protocol 023) was a multi-center, randomized, double-blind, placebo-controlled, Phase II test-of-concept clinical trial. The HIV-negative participants in this trial were from multiple clinical trial sites in North and South America, the Caribbean and Australia, where HIV subtype B, the subtype of HIV from which the HIV genes included in the vaccine are derived, is predominant. When injections were halted, 2,677 study participants had received all three doses of vaccine or placebo.
A second Phase II test-of-concept trial of the Merck T-cell vaccine candidate, the Phambili study, was started in 2007 in South Africa by the HVTN to explore whether Merck's vaccine would be effective at preventing infection, reducing viral levels, or both, from HIV subtype C, which is more common in southern Africa and many other parts of the world with the highest rates of new HIV infections. Fifty-six volunteers received three doses of the vaccine or placebo. Enrollment of participants in the Phambili study was interrupted when the early results from the Step trial showed lack of efficacy.
Step Study Results
The Step study evaluated two primary efficacy endpoints: whether the vaccine prevented HIV infection in those who were HIV negative at the start of the study, and whether the vaccine reduced the amount of virus in those who became HIV infected during the course of the study. These primary efficacy analyses were based on volunteers who had low levels of pre-existing immunity to adenovirus type 5 (Ad5), the cold virus used as a carrier for the synthetic HIV genes in the vaccine. Results showed:
-- 24 cases of HIV infection were observed in the 741 volunteers who had received at least one dose of the vaccine, and 21 cases of HIV infection were observed in the 762 participants in the placebo group.
-- HIV RNA levels approximately 8 to 12 weeks after diagnosis of infection were generally similar in the vaccine and the placebo groups. The geometric means of the HIV RNA levels in the blood of infected individuals, the standard measure of ongoing HIV replication, were approximately 40,000 copies/mL in the 24 volunteers in the vaccine group who developed HIV infection and approximately 26,000 copies/mL in the 21 volunteers in the placebo group who developed infection.
Extensive additional analyses have been and continue to be done to better understand the results from the Step study. In exploratory post-hoc analyses, HIV incidence was higher in those men who had high levels of pre-existing immunity to Ad5 (5.1 percent in the vaccine group versus 2.2 percent in the placebo group per year) and those who were uncircumcised (5.2 percent in vaccine group versus 1.4 percent in placebo group per year).
Further information is available on the HVTN website: www.hvtn.org/
Source: Fred Hutchinson Cancer Research Center