New sugar-based catalyst could offer a potential solution for using captured carbon
A new catalyst made from an inexpensive, abundant metal and common table sugar has the power to destroy carbon dioxide (CO2) gas.
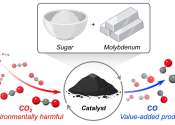
A new catalyst made from an inexpensive, abundant metal and common table sugar has the power to destroy carbon dioxide (CO2) gas.
Analytical Chemistry
May 2, 2024
0
17

In most industrial chemical reactions, catalysts combine with the starting materials and accompany them through intermediate stages to the product. In chemistry, this pathway is known as the reaction mechanism, and it is ...
Analytical Chemistry
Apr 25, 2024
0
14

Gas sensors have been widely applied in areas such as medical health, environmental monitoring, and food safety. However, current gas sensors still face several challenges, including low sensitivity, long response and recovery ...
Analytical Chemistry
Apr 22, 2024
0
1

There is a high level of interest, even excitement, among chemists and materials scientists about the potential of single-atom catalysts (SACs), but their development relies on very specialized tools available only at synchrotrons ...
Analytical Chemistry
Apr 22, 2024
0
14

Since the advent of industrial revolution, the accumulation of carbon dioxide (CO2) in the Earth's atmosphere has raised significant environmental and climate concerns. As a response to this pressing challenge, the conversion ...
Analytical Chemistry
Apr 22, 2024
0
0

Researchers at the University of Twente have shown how to improve the efficiency of hydrogen production in an experimental setup. They showed that the magnetic order of the molecules plays a critical role.
Condensed Matter
Apr 16, 2024
0
20

Alcohols are widely applied in life sciences and the chemical industry. Selective hydrogenation of epoxides using hydrogen molecules as a reductant is considered to be one of the most facile and atom-economical strategies ...
Analytical Chemistry
Apr 15, 2024
0
8

Fuel cells are quickly becoming a viable, clean energy alternative to commonly used fossil fuels, such as gasoline, coal, and oil. Fossil fuels are non-renewable energy resources that release carbon dioxide into the atmosphere.
Analytical Chemistry
Apr 10, 2024
0
50

Researchers at The University of Manchester have developed a new catalyst which has been shown to have a wide variety of uses and the potential to streamline optimization processes in industry and support new scientific discoveries.
Analytical Chemistry
Apr 8, 2024
0
30

The active ingredient in many drugs is what's known as a small molecule: bigger than water, much smaller than an antibody and mainly made of carbon. It's tough, however, to make these molecules if they require a quaternary ...
Materials Science
Apr 6, 2024
4
81
Catalysis is the process in which the rate of a chemical reaction is either increased or decreased by means of a chemical substance known as a catalyst. Unlike other reagents that participate in the chemical reaction, a catalyst is not consumed by the reaction itself. The catalyst may participate in multiple chemical transformations. Catalysts that speed the reaction are called positive catalysts. Catalysts that slow down the reaction are called negative catalysts or inhibitors. Substances that increase the activity of catalysts are called promoters and substances that deactivate catalysts are called catalytic poisons. For instance, in the reduction of ethyne to ethene, the catalyst is palladium (Pd) partly "poisoned" with lead(II) acetate (Pb(CH3COO)2). Without the deactivation of the catalyst, the ethene produced will be further reduced to ethane.
The general feature of catalysis is that the catalytic reaction has a lower rate-limiting free energy change to the transition state than the corresponding uncatalyzed reaction, resulting in a larger reaction rate at the same temperature. However, the mechanistic origin of catalysis is complex. Catalysts may affect the reaction environment favorably, e.g. acid catalysts for reactions of carbonyl compounds, form specific intermediates that are not produced naturally, such as osmate esters in osmium tetroxide-catalyzed dihydroxylation of alkenes, or cause lysis of reagents to reactive forms, such as atomic hydrogen in catalytic hydrogenation.
Kinetically, catalytic reactions behave like typical chemical reactions, i.e. the reaction rate depends on the frequency of contact of the reactants in the rate-determining step. Usually, the catalyst participates in this slow step, and rates are limited by amount of catalyst. In heterogeneous catalysis, the diffusion of reagents to the surface and diffusion of products from the surface can be rate determining. Analogous events associated with substrate binding and product dissociation apply to homogeneous catalysts.
Although catalysts are not consumed by the reaction itself, they may be inhibited, deactivated or destroyed by secondary processes. In heterogeneous catalysis, typical secondary processes include coking where the catalyst becomes covered by polymeric side products. Additionally, heterogeneous catalysts can dissolve into the solution in a solid-liquid system or evaporate in a solid-gas system.
This text uses material from Wikipedia, licensed under CC BY-SA